Atoms
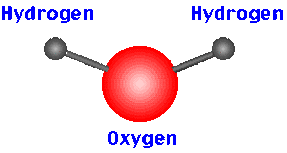
Atoms are the fundamental building blocks of almost everything, from a speck of dust to the biggest galaxies out in the universe. They make up the entire universe! Even you and I are made up of atoms. Atoms can be combined to make molecules. For example, if 2 hydrogen atoms are combined with one oxygen atom, H2O, or water, will be formed. Atoms are so small that approximately 50 million atoms of solid matter lined up in a row only measure 1cm!
Structure
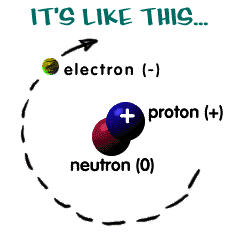
If we look inside an atom, we will see that there are electrons, protons, and neutrons. The electrons are "orbiting" around the nucleus, which consists of protons and neutrons, of the atom. The nucleus of an atom is so small, its volume is only 1/10,000 of the volume of the atom. This means that atoms are mostly empty space. If you take a look at your desk or your hand, 99.99% of it is empty space! It's quite hard to believe that everything you touch is mostly free space. But won't we just go through everything since there is so much empty space? The answer is no. The reason is that the electrons repel each other strongly, just like two magnets with same poles.
Elements, Isotopes, and the Periodic Table
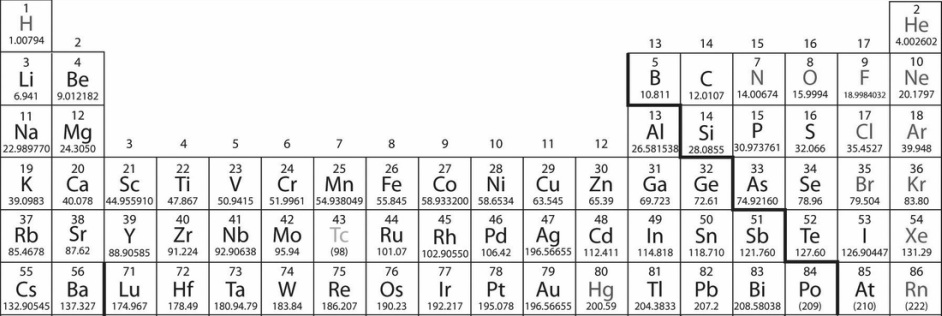
Here is a part of the periodic table. Some elements you may recognize, such as Hydrogen(H) and Oxygen(O). Have you ever wondered what the numbers above and below the elements actually represent? The number above represents the number of protons. For example, Lithium(Li) has 3 protons. The number on the bottom is the average of all the masses of isotopes of the element. What's an isotope? It is a variant of the same element. An element and its isotope have the same number of protons and electrons but they have different number of neutrons. Therefore, the mass changes in the isotopes. An example of an isotope is deuterium or H2. Hydrogen has no neutrons but deuterium has 1 neutron. So deuterium is heavier than hydrogen is sometimes called heavy hydrogen.
